Similarly, a decrease in solute concentration decreases the specific gravity of urine, which indicates medical problems like renal failure. An increase in the specific gravity of urine indicates that it is due to an increase in the solutes caused by dehydration, diarrhea, or infection. Both high and low density or specific gravity than the normal range of urine indicates medical problems. Both the blood and urine have dissolved substances in water that increase the density from that of pure water. The density of urine varies in the range of 1.0050 g/mL to 1.030 g/mL. For example, the density of whole blood for humans is ~1.060 g/mL. Water (chemical formula: H2O) is a transparent fluid which forms the worlds streams, lakes, oceans and rain, and is the major constituent of the fluids of organisms. Handbook Of Chemistry and Physics Online. It is an oxygen hydride, a mononuclear parent hydride and an inorganic hydroxy compound. Calculate the density of a liquid in a beaker. Click the help icon for more information on the differences between Individual User accounts and Workspace accounts. If you know that the object has a density of 20.0 g/ml and the volume is 5.00 ml. 
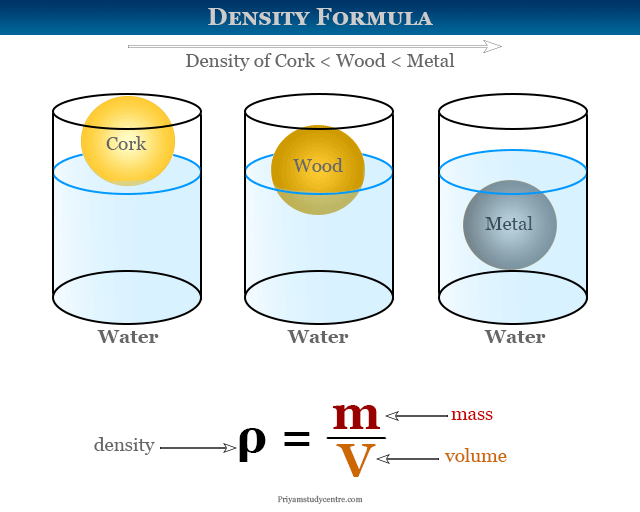
Density determines what floats and what sinks. 
When substances dissolve in water, the density of the solution is usually different from pure water. Cold Water Density Movie 2 Purdue: Density- The proportion of mass to volume of an object. The density of water is 1.0 g/mL at room temperature, so the specific gravity is equal to the density of the object expressed without a unit. Solubilities are in water and are reported as grams solvent/100 grams water. The values were obtained from the CRC (87th edition), or Vogels Practical Organic Chemistry (5th ed.). -to-Grams-(g)-Step-12.jpg)
Therefore, the specific gravity is a unitless number. The gram per cubic centimetre is a unit of density in the CGS system, and is commonly used in chemistry.It is defined by dividing the CGS unit of mass, the gram, by the CGS unit of volume, the cubic centimetre.The official SI symbols are g/cm 3, g·cm 3, or g cm 3.It is equivalent to the units gram per millilitre (g/mL) and kilogram per litre (kg/L). Myers, Webmaster of ACS Division of Organic Division (DOC) from: Professor Murovs Organic solvent table. So, #"2.46 mL"# of water at a temperature of wil contain a total of #8.23 * 10^(22)# molecules of water.\nonumber #2.46color(red)(cancel(color(black)("g"))) * "1 mole water"/(18.015color(red)(cancel(color(black)("g")))) = "0.1366 moles water"#įinally, the relationship between number of moles and number of molecules is given by Avogadro's number, which tells you that every mole of a substance contains exactly #6.022 * 20^(23)# molecules of that substance. This tells you that every mole of water has mass of #"18.015 g"#. This means that #"2.46 mL"# will have a mass of Actually, the exact density of water is not really 1 g/ml, but rather a bit less (very, very little less), at 0.9998395 g/ml at 4.0 Celsius (39.2 Fahrenheit). End result: 100 ml is equal to 104.166667 g. A common unit of measurement for water's density is gram per milliliter (1 g/ml) or 1 gram per cubic centimeter (1 g/cm 3). It is known that the density of vegetable oils is about 0.96 grams per ml. In this case, a density of #"`1.00 g/mL"# tells you that every milliliter of water has a mass of #"1.00 g"#. Sample task: convert 100 milliliters of vegetable oil to grams. So, density is defined as mass per unit of volume. The idea here is that you need to use the density of water at and the volume of the sample to find its mass, then use water's molar mass to find how many moles of water you get in this sample. Sample task: convert 500 grams of water to milliliters, knowing that the density of water is one gram per milliliter. Pure Water Density Standard UKAS ISO/IEC17025 and ISO Guide 34 certified, density: 0.9982 g/mL at 20 ☌, density: 0. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
